Silicon
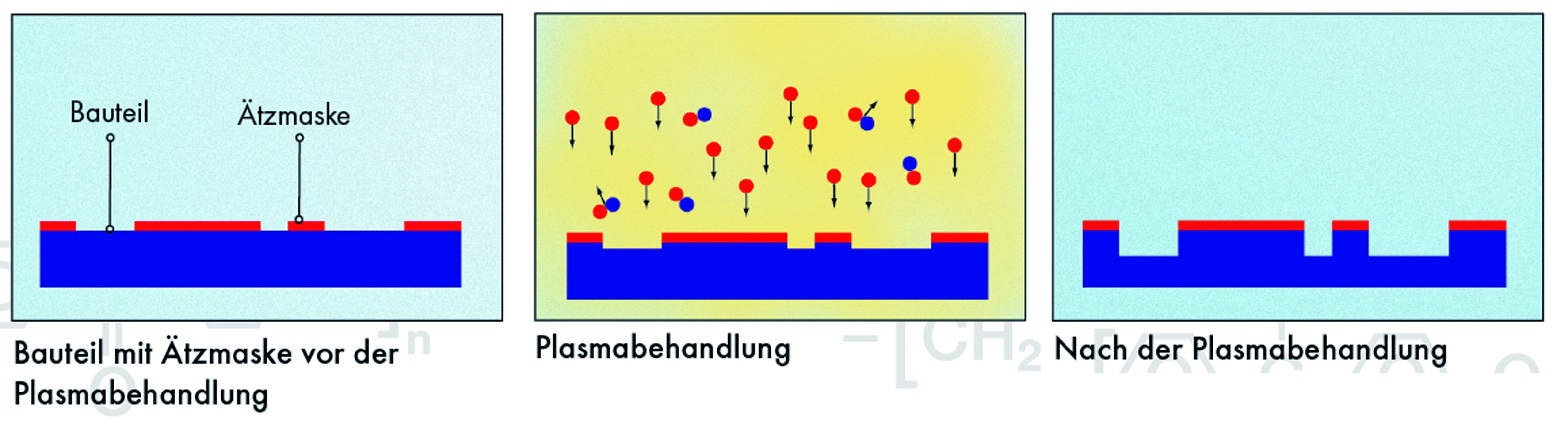
Silicon is one of the most important elements, but not because of or since the triumph of microelectronics. Silicon accounts for about 15% of the total mass of the earth, but in nature it occurs only as a compound, especially as a mineral compound and primarily as silicon dioxide SiO2. This mineral is the main component of granite, sand, and sandstone. The pure SiO2 mineral is referred to as quartz. In pure form, it can also be processes as silica glass. Generally, it is the main component of all glass. Silicon has the atomic number 14; in the periodic table, it is in the carbon group. With its 4 valence electrons, it has multiple ways of forming compounds and crystal structures, comparable to carbon. In its elementary form, silicon is a semi-metal or semiconductor and the most important element for commercial applications. Here, its capability of forming very large disturbance-free monocrystals is of particular significance. Many methods of silicon handling are implemented by plasma engineering, in particular structuring by means of etching. Fluorine compounds such as sulphur hexafluoride SF6 or tetrafluormethane CF4 are often used as etching gases. Furthermore, photolithography processes are used to apply conductors or protective coats by means of plasma polymerisation.