
Wetting
Wetting is present if a liquid applied to solid dissolves. This is not necessarily the case. Liquids have a surface tension which results from the mutual bonding forces of the surface molecules. Due to this surface tension, a volume of liquid always wants to take on a spherical shape. Only if the forces of attraction between the molecules, the surface of the solid body and the liquid molecules are higher than the forces which keep the shape of the liquid surface in form, does the liquid dissolve on the solid surface, i.e. wetting occurs. In general, the following applies: For wetting to occur, the surface energy of the solid body must be higher than the surface tension of the liquid. To ensure that a substance adheres to a surface, wetting of the surface by the substance must take place. Accordingly, if you wish to bond, print or paint a surface, the adhesive, paint or ink must wet the surface. Plasma technology offers a wide range of options for effectively modifying the surface energy of solid bodies, and thus their wettability. For more information, refer to the section⇒ Wettability
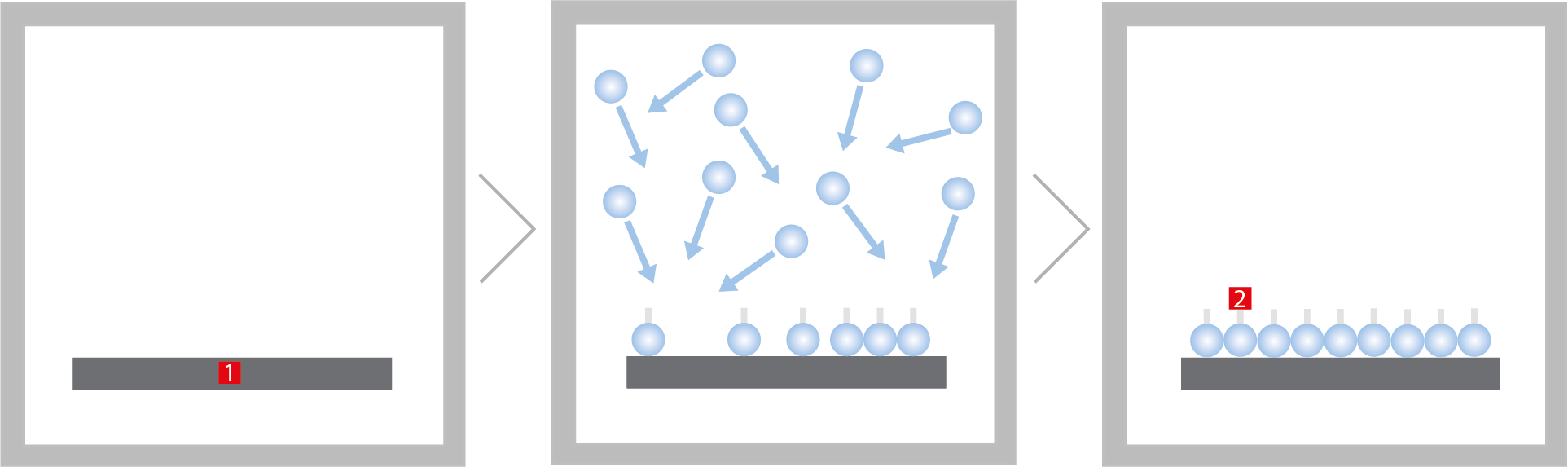
Functional principle of a plasma activation

Oxygen
