Glossary of surface technology
Plasma cleaning
Before any further plasma treatment can be done, plasma cleaning must be carried out first to make a pure material surface available. For complete cleaning, choose one of the following processes:
Cleaning in the oxygen plasma
Surface contaminations, in particular the residues remaining after mechanical and wet-chemical cleaning, are often organic. In many cases, these are residues of oils, greases, release agents, and silicones which many solvents cannot remove completely. If these substances remain on the surface, they impair all downstream processing steps and have a particularly negative effect on all types of bonding and coating. In most cases, these substances can be removed residue-free in an oxygen plasma, often also an air plasma. meist restlos entfernt werden. Excited oxygen molecules and radicals are highly reactive and form very stable bonds.The UV radiation of the radiation splits the polymer chains of the organic residues. Oxygen radicals immediately occupy the released bonds and prevent the polymer fragments from re-uniting. In this way, short-chain volatile substances which can be sucked off by the vacuum pump are increasingly formed from the macromolecules.
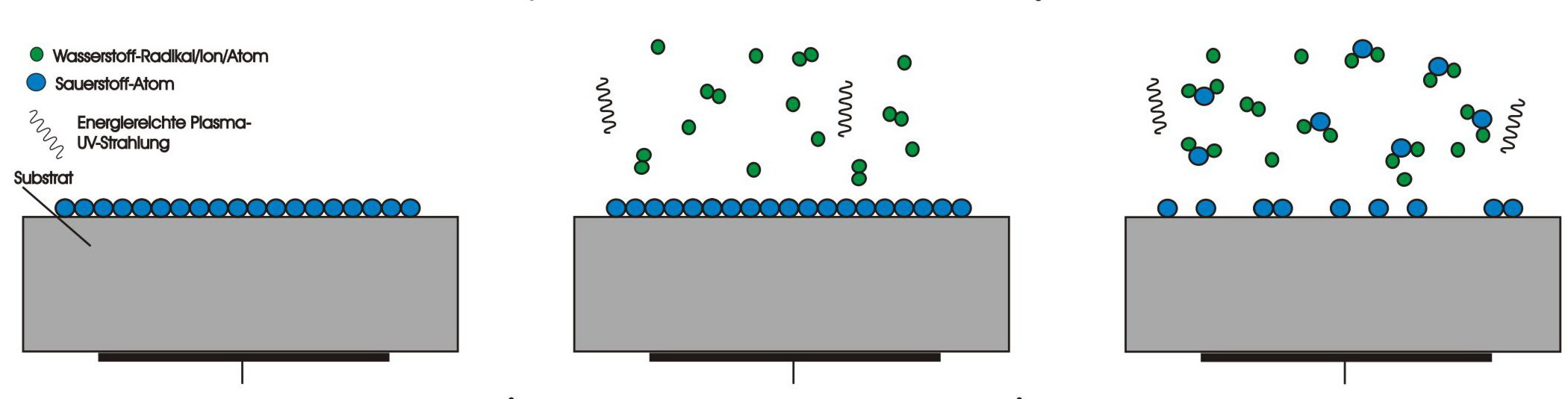
Decomposition of oxide layers
On the surface of almost all metals, an oxide layer forms even if the metal is only briefly exposed to the air atmosphere. This effect is extremely useful for many base metals, as the usually solid, resistant oxide layer prevents corrosion of the metal beneath it. However, oxide layers impede subsequent joining processes, especially soldering and bonding, as well as electrical contacting. In the hydrogen plasma, excited hydrogen molecules, ions and radicals react with the oxygen of the oxide to form vapour which is easily extracted by the vacuum pump.Micro-sandblasting in the argon plasma
Some substances can be removed neither by oxygen plasma nor by hydrogen plasma , in particular salts and ceramic substances. In these cases, physical etching by ion bombardment in the argon plasma is used, i.e. atoms, radicals and molecules are broken off a surface by the kinetic energy of the ion bombardment. This effect is not selective, which means that it works on practically all substrates. Thus, virtually all substances can be removed by sufficiently intensive argon plasma treatment. However, the argon ion bombardment will also etch the substrate itself and remove substrate particles. This may be desirable for the roughening effect which, as with sandblasting or grinding, increases the area and improves the bonding in gluing or coating processes. If substrate etching is not desirable, the treatment time in the argon plasma must be optimally adjusted to prevent it.The processing speed with micro-sandblasting is low. For this reason, initial cleaning in an oxygen plasma takes place to remove hydrocarbons, if applicable. Next, argon plasma treatment can follow. When oxide layers must be removed as well, an argon/hydrogen process gas can be used for cleaning.