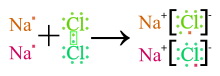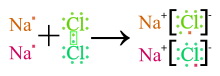Ionic bond
Bond based on the electrostatic attraction between anions and cations. Generally the strongest form of molecule bond. It is present in bonds between atoms with highly different electronegativity. This is particularly applicable for the bond between atoms of the first main group, i.e. with an electron at the highest energy level and atoms of the seventh main group with exactly one electron hole at the highest energy level. In this bond, the atom of the 1st main group very easily releases its sole electron to the hole in the atomic shell of the atom of the 7th main group. This makes the atom of the 1st main group a positively charged ion (cation), and the atom of the 7th main group a negatively charged ion (anion).